You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
What is Thermodynamics: Definition and 1000 Discussions
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, radiation, and physical properties of matter. The behavior of these quantities is governed by the four laws of thermodynamics which convey a quantitative description using measurable macroscopic physical quantities, but may be explained in terms of microscopic constituents by statistical mechanics. Thermodynamics applies to a wide variety of topics in science and engineering, especially physical chemistry, biochemistry, chemical engineering and mechanical engineering, but also in other complex fields such as meteorology.
Historically, thermodynamics developed out of a desire to increase the efficiency of early steam engines, particularly through the work of French physicist Nicolas Léonard Sadi Carnot (1824) who believed that engine efficiency was the key that could help France win the Napoleonic Wars. Scots-Irish physicist Lord Kelvin was the first to formulate a concise definition of thermodynamics in 1854 which stated, "Thermo-dynamics is the subject of the relation of heat to forces acting between contiguous parts of bodies, and the relation of heat to electrical agency."
The initial application of thermodynamics to mechanical heat engines was quickly extended to the study of chemical compounds and chemical reactions. Chemical thermodynamics studies the nature of the role of entropy in the process of chemical reactions and has provided the bulk of expansion and knowledge of the field. Other formulations of thermodynamics emerged. Statistical thermodynamics, or statistical mechanics, concerns itself with statistical predictions of the collective motion of particles from their microscopic behavior. In 1909, Constantin Carathéodory presented a purely mathematical approach in an axiomatic formulation, a description often referred to as geometrical thermodynamics.
View More On Wikipedia.org
Historically, thermodynamics developed out of a desire to increase the efficiency of early steam engines, particularly through the work of French physicist Nicolas Léonard Sadi Carnot (1824) who believed that engine efficiency was the key that could help France win the Napoleonic Wars. Scots-Irish physicist Lord Kelvin was the first to formulate a concise definition of thermodynamics in 1854 which stated, "Thermo-dynamics is the subject of the relation of heat to forces acting between contiguous parts of bodies, and the relation of heat to electrical agency."
The initial application of thermodynamics to mechanical heat engines was quickly extended to the study of chemical compounds and chemical reactions. Chemical thermodynamics studies the nature of the role of entropy in the process of chemical reactions and has provided the bulk of expansion and knowledge of the field. Other formulations of thermodynamics emerged. Statistical thermodynamics, or statistical mechanics, concerns itself with statistical predictions of the collective motion of particles from their microscopic behavior. In 1909, Constantin Carathéodory presented a purely mathematical approach in an axiomatic formulation, a description often referred to as geometrical thermodynamics.
View More On Wikipedia.org
-
B
Heat distribution in a piece of glass receiving protons
First of all, I didn't know whether to pick this subforum or the engineering/compsci one, I understand this might need to be moved to a more appropriate subforum. The general approach is fairly obvious, use implicit method to construct the tridiagonal matrix for Thomas method and solve. However...- Bqback
- Thread
- Replies: 6
- Forum: Advanced Physics Homework Help
-
J
The Physics of cooking breakfast (basic thermodynamics)
I eat eggs, ham, peppers, and a pancake for breakfast. I cook the ham and peppers at the same time. For purposes of this question, I have the eggs and the pancake figured out. Given a gas stove, a small, nonstick aluminum frying pan (with lid) and corn oil; If my goals are (in order of...- jamesson
- Thread
- Replies: 1
- Forum: Thermodynamics
-
A
Gas - cylinder - piston problem
Summary:: Seeking explanation to classical gas - cylinder - piston problem, not the solution. Problem 1.15 from 7th edition of Introduction to Chemical Engineering Thermodynamics by Smith, Van nes and Abbot) Classical problem, given: - gas in a confined cylinder - piston with weight is placed...- akmkeng
- Thread
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-
T
Thermodynamics -- Calculations for heating water up
A thermal flask with water of temperature 90c is placed out in room temperature 20c, sealed. How long before it reaches 55c? outer radius and height: 5cm, 25cm volume of water is 1litre. R = 0.5 Km2/W using this equation to solve it combining these two and integrating with respect to T...- tollf
- Thread
- Replies: 2
- Forum: Introductory Physics Homework Help
-
Y
Why use c_p and not c_v as specific heat - Thermodynamics
Hey all, I am working on a problem that goes like this: The cargo space of a refrigerated truck whose inner dimensions are 12 m 3 2.3 m 3 3.5 m is to be precooled from 25°C to an average temperature of 5°C. The construc- tion of the truck is such that a transmission heat gain occurs at a rate...- yjl
- Thread
- Replies: 2
- Forum: Introductory Physics Homework Help
-
I
Question about thermal physics -- Ice cubes melting in water
First, I calculated the heat required for the ice to melt: Q=mLf Q=0.150×330 Q=49.5 J Then, I calculated the final temperature of the water by forming the following equation: Q=mcΔT −49.5=(0.15+0.35)×4200×(Tf −80) Tf=80.0 degrees Celcius But the answer says 32 degrees Celsius.- ianc1339
- Thread
- Replies: 8
- Forum: Introductory Physics Homework Help
-
H
I Thermodynamics Differentiating logarithms
<mentor: change title> In thermodynamics, there is a function which, for the three variables x, y, and z, can be given as ##G = xG_x+yG_y+zG_z + N[x\ln(x) + y\ln(y)+ z\ln(z)]+E(x,y,z)## where G_x, G_y, G_z, and N are some constants and E is some arbitrarily complicated term. There is a...- Hypatio
- Thread
- Replies: 11
- Forum: Differential Equations
-

Impossible thermodynamics Q :(((
- RoisinCleary
- Thread
-
- Tags
- Impossible Thermodynamics
- Replies: 1
- Forum: Introductory Physics Homework Help
-
A
Conceptual thermodynamics problem about ammonia executing a Carnot cycle
Is there a mathematical explanation for why the work done in the condenser (in process 2 to 3) is zero? I am aware that ammonia does not expand or compress in the condenser, only changes phase, but without knowing that the process takes place in a condenser and only considering the graph...- Andrew1234
- Thread
- Replies: 3
- Forum: Introductory Physics Homework Help
-
A
Conceptual thermodynamics question regarding specific heat ratio
The solution can be found at https://study.com/academy/answer/an-insulated-rigi... After using the two equations I can't see why (h2-h1)/(u2) should equal (T2)/(T1). Can someone explain why specific heat ratio is equal to temperature ratio?- Andrew1234
- Thread
- Replies: 6
- Forum: Introductory Physics Homework Help
-

The Second Law of Thermodynamics and the Stefan-Boltzmann Law
I have a question about the Second Law of Thermodynamics and the Stefan-Boltzmann law. These quotes are from http://www.ces.fau.edu/nasa/module-2/correlation-between-temperature-and-radiation.php “The Stefan-Boltzmann law, a fundamental law of physics, explains the relationship between an...- jackdale
- Thread
- Replies: 6
- Forum: Thermodynamics
-
P
Thermodynamics Problem: Reversible and Irreversible Processes
Attempt at A Solution Problem 1 Reversible Process - A cylinder of ideal gas at pressure P is in mechanical equilibrium with a piston of area A driven by a spring of force F = PA and thermal equilibrium with a reservoir of temperature T. The piston is moved a small distance dx toward the...- Pendleton
- Thread
- Replies: 1
- Forum: Introductory Physics Homework Help
-

Fundamental equation (thermodynamics) from Euler's equation
by substituting all values in the euler equation you get most of the terms in the fundamental equation but not (N/No)^-(c+1) How do you get this term?- VVS2000
- Thread
- Replies: 1
- Forum: Thermodynamics
-
M
Thermodynamics Problem: Cylinder-Piston System with Friction
Hi guys, First of all I'm sorry for my bad english I'll try to be as clear as possible. I have tried to solve this problem to understand the First Law of Thermodinamics: Q+L=ΔE_t In fact I know L (in the current convention) is the work which the envirorment does on the system but I don't...- MathError
- Thread
-
- Tags
- Friction System Thermodynamics
- Replies: 4
- Forum: Introductory Physics Homework Help
-
S
High Energy Introductory notes on AdS/CFT or black hole thermodynamics
I am looking for a book/notes on the topics mentioned in the title that would be accessible to an undergrad. I have a background in grad quantum and statistical mechanics, but most resources I found on those topics assume a familiarity with QFT, string theory, gauge theory, and general...- Silicon-Based
- Thread
- Replies: 2
- Forum: Science and Math Textbooks
-
B
Thermodynamics: calculate thermodynamic derivative from data?
I don't understand how to use output from an NPT molecular dynamics simulation to compute a thermodynamic derivative. I need to compute this (where "d" is a partial derivative, "T" is a subscript that means, "at constant temperature," and "E" is internal energy): -(dE/dV)T I have a simulation...- bumblebee77
- Thread
- Replies: 1
- Forum: Thermodynamics
-

Integration of inexact differentials in Thermodynamics
In Thermodynamics, I have seen that some equations are expressed in terms of inexact differentials, ##\delta##, instead of ##d##. I understand that this concept is introduced to point out that these differential forms are path-dependent, although I am not clear how they can be handled. So, are...- Sokolov
- Thread
- Replies: 1
- Forum: Thermodynamics
-
Y
Engineering Find out the percent exergy loss
Problem, with state values, and pie chart (Fig 4.20) showing answers: ^ This shows the system in question (Kapitza Liquefaction System). Methane gas enters into the compressor (c), then goes through the first heat exchanger (HX1). Some of it (z) gets routed to the expander (exp). Afterwards...- yaro99
- Thread
- Replies: 17
- Forum: Engineering and Comp Sci Homework Help
-
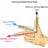
Engineering Thermodynamics - Control Volumes find final temperature
Hi I'd like to know if my current approach to the problem has any issues. My attempt: Balancing the equation: Qin + Win +mΘout = Qout + Wout + mΘout Qin+m(hin + v2in/2 + gzin) = m(hout + v2out/2 + gzout) Wflow + ΔU + m(hin) = m(hout) I factored out the mass: wflow + Δu + hin = hout And I used...- WateryFungi
- Thread
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-
A
Thermodynamics energy balance for control volume
Why is energy balance for a control volume dE/dt = dQ/dt-dW/dt-dm/dt(ΔH+ΔKE+ΔPE) 0 = dQ/dt-dW/dt-dm/dt(ΔH+ΔKE+ΔPE) whereas for other systems it is ΔE =Q-W-(ΔU+ΔKE+ΔPE) 0 = Q-W-(ΔU+ΔKE+ΔPE) with enthalpy, h = u +pv, replaced by only the internal energy? How is the pv term accounted for...- Andrew1234
- Thread
- Replies: 1
- Forum: Introductory Physics Homework Help
-
M
Engineering Thermodynamics - Second Law: 2 Heat Engines Connected Between 3 Metal Blocks
Hi, I posted a similar question recently and gained some insight on these types of problems. However, I am slightly stumped on how to approach this variation of the problem. So I know that: - there is no net change in enthalpy of the blocks and the engine as the processes are reversible -...- Master1022
- Thread
- Replies: 9
- Forum: Engineering and Comp Sci Homework Help
-
M
Thermodynamics Problem: Heat Engine Between Two Blocks
Hi, I am quite confused about how to approach this problem. I have seen variations of this problem where there is a heat engine between two blocks, but in this case the surroundings are massless, so I don't believe that approach will work here. Method: I have first started with the case that...- Master1022
- Thread
- Replies: 16
- Forum: Introductory Physics Homework Help
-

Isothermal pressure change in a U-shaped tube
Hi, just reviewing some thermodynamics from the textbook by Sears and Salinger, having a hard time conceptualizing this one. It's an isothermal change in pressure, so the volumes of the mercury and the air both change to reach equilibrium, but if it's a "good vacuum pump", then won't the right...- obstinatus
- Thread
- Replies: 4
- Forum: Introductory Physics Homework Help
-
C
Thermodynamics help please -- Air passing through a gas turbine system
Summary:: NO TEMPLATE BECAUSE THIS HOMEWORK PROBLEM WAS MISPLACED IN A REGULAR FORUM Cant do part c, using the steady flow equation I am confused how to continue. Please help! Mainly confused as to what heat transfer loss represents in the steady flow equation and where to go to find the...- clark123
- Thread
- Replies: 17
- Forum: Engineering and Comp Sci Homework Help
-

Where do I put this Server Room's air exhaust?
Hey guys. I have a room which i want to use as a server room. the devices need to work 24/7 and they get pretty hot. so it is imperative to keep the room cool otherwise the devices will be damaged. I have an air vent to bring cold air into the room for the devices so their fans can suck in cold...- Anon_Miner
- Thread
- Replies: 15
- Forum: Mechanical Engineering
-

Trouble with fluid thermodynamics and nuclear thermal rockets
Summary:: In need of help determining the exhaust velocity of a rocket nozzle given temperature and propellant molar mass Greetings and salutations! My name is Robert DeVries, world builder extraordinaire. I have come with questions in search of answers. So for the last few days I've been...- Robert DeVries
- Thread
- Replies: 2
- Forum: Sci-Fi Writing and World Building
-
Z
Thermodynamics: find the change of internal energy, the work and Q
According to the first principle of thermodynamics: ΔU = W + Q Also noting that: W = -P⋅ΔV (Question: This P is the initial pressure or the final?) To find V2: (P1⋅V1) / T1 = (P2⋅V2) / T2 → Therefore, (P⋅V1) / T1 = [(P/5)⋅V2] / T2 → (P⋅V1) / T1 = (P⋅V2) / (5⋅T2) → V2 = (5⋅T2⋅V1) / T1...- zvwner
- Thread
- Replies: 10
- Forum: Introductory Physics Homework Help
-

Thermodynamics -- Temperature of a Heat Source?
In heat engine we define a heat source from where heat is transferred to the system, we say that heat source has a temperature ##T_h## , When we define a Carnot heat engine, the first process we have is an isothermal expansion and we say heat has to come in system through this process and here...- Rahulx084
- Thread
- Replies: 16
- Forum: Materials and Chemical Engineering
-
T
1st Law of thermodynamics : moving reference frame
I'd like to apply the 1st law of thermodynamics in a reference frame (RF) moving with constant velocity. We have: ##\Delta{}E = E_{in} - E_{out}## I am limiting myself to rectilinear motion. Suppose we are in a RF moving with a constant velocity ##V##. Let the system consist of a mass ##m##. The...- thinkingcap81
- Thread
- Replies: 8
- Forum: Thermodynamics
-
E
Confused about use of external vs internal pressure in thermodynamics
I've learned that ##W = -\int{P_{ext} dV}##, and only during a reversible/quasi-static process where ##P_{int} = P_{ext}## can we write the work done on the gas in terms of the internal pressure (and consequently use ##PV=nRT## etc. which apply to the internal gas). However, a lot of sources...- etotheipi
- Thread
- Replies: 13
- Forum: Thermodynamics
-
E
Ignoring the acceleration of a piston in thermodynamics
I was just reading a set of thermodynamics lecture notes and came across the following In most thermodynamics problems I have done, it is indeed assumed that the piston does not accelerate so we can simply equate forces on the piston. However, I don't fully understand the line of reasoning...- etotheipi
- Thread
- Replies: 4
- Forum: Thermodynamics
-
C
B Irreversible expansion of gas against gas
Let's say I have a liter of gas at pressure of 4 atm and T=900K. I use it to move perfect massless frictionless and insulative piston to compress a liter of 1 atm, T=300K gas. When the pressure on both sides is equal and the piston stops moving, will the temperature on both sides of the piston...- Christofer Br
- Thread
- Replies: 10
- Forum: Classical Physics
-
T
Adiabatic approximation in the derivation of the speed of sound
The speed of sound in a gas at temperature T is given to be ## v=\sqrt{\frac{\gamma RT}{M}}##, where ##\gamma## is the adiabatic exponent, R is the gas constant and M is the molar mass of the gas. In deriving this expression, we assumed that the compression and expansion processes were so fast...- Terry Bing
- Thread
- Replies: 6
- Forum: Mechanics
-
F
Temperatures of the hot source and cold sink in a heat engine
Hi, I was just wondering about the efficiency of a cycle that is not Carnot cycle. In that case one should use \eta = 1-\left|\frac{Q_{\rm out}}{Q_{\rm in}}\right|, where Q_{\rm in} and Q_{\rm out} are the amounts of heat absorbed and released during the cycle. For instance, I guess that in...- FranzDiCoccio
- Thread
- Replies: 2
- Forum: Thermodynamics
-
H
Thermodynamics: Why does salt lower the melting point of ice?
There was a question on "Why salt lower the freezing point of water?"I found the following answer."Thermodynamics teaches that a loss of entropy can be overcome by a gain in so called enthalpy". The loss of entropy by freezing the solution canbe over come at temperature much below 0 degree C...- hariharan venkatasu
- Thread
- Replies: 21
- Forum: Chemistry
-
C
Thermodynamics - Change in temperature for heated slabs of steel
Just started this topic so I'm not sure if this is the correct way to solve this, any help would be appreciated. mass of object x change in temp x specific heat capacity= heat change in temp= heat/(mass of object x specific heat capacity) Mass= 700kg Specific heat capacity of steel= 0.42kJ/kgC...- CJoy
- Thread
- Replies: 3
- Forum: Introductory Physics Homework Help
-

Thermodynamics problem involving engine efficiency
Q2: An engine has power of 26.5 kW, needs 9 kg coal during an hour for energy. The heat capacity of coal is 7800 cal/g. Define the engine’s efficiency. Qh= mcΔT = 9000x7800xΔT ( stuck) P=26.5kW(is this the power output of the engine in an hour?) If only I could find ΔT, thenI would be able to...- Stephen Bulking
- Thread
- Replies: 20
- Forum: Introductory Physics Homework Help
-

Finding Isentropic Enthelpy, knowing Isentropic Entropy
A short background: My question focuses solely on the part of the refrigeration cycle to do with the compressor, where the cycle begins. The first state is before the refrigerant enters the compressor, and the second state is after the refrigerant leaves the compressor. My goal is to obtain...- WhiteWolf98
- Thread
- Replies: 3
- Forum: Introductory Physics Homework Help
-
A
Does classical statistical physics predict newer things vs. thermodynamics?
I'm wondering if the passage from a classical thermodynamic theory, i.e. which does not resort to an atomistic theory and methods of probability and statistics, to classical (i.e. non-quantum) statistical mechanics, led to new discoveries and especially if it was able to explain properties of...- Aidyan
- Thread
- Replies: 4
- Forum: Thermodynamics
-

Entropy and the Laws of Thermodynamics
Hello everyone, I have to write a paper about entropy and how it relates to the laws of thermodynamics, energy, and work. I have taken a deductive approach starting from the zero-th law to the second law of thermodynamics as follows. Entropy is the disorder of a system (Class Video, 2019)...- Physics345
- Thread
-
- Tags
- Entropy Laws Thermodynamics
- Replies: 3
- Forum: Introductory Physics Homework Help
-
C
Is Specific impulse indicative of performance as a gun propellant?
Black powder has specific impulse of around 80s, while rocket candy has up to 130s of specific impulse. Does that mean I could replace the propellant in a BP cartridge with 80/130 of the weight in rocket candy and obtain the same performance in an idealized gun? (as in without considering...- Christofer Br
- Thread
- Replies: 1
- Forum: General Engineering
-
D
Entropy and the second law of thermodynamics
- denniszhao
- Thread
- Replies: 13
- Forum: Introductory Physics Homework Help
-

Clausius-Clapeyron equation to estimate sublimation pressure of water
Before this question, the questions were about the Clapeyron equation, and how to estimate ##\Delta s##. I'm completely put off by this question however, and don't know where to start. I've found that the triple point of water is at ##0.01°C##, and there is indeed data in the table for...- WhiteWolf98
- Thread
- Replies: 5
- Forum: Introductory Physics Homework Help
-
K
Gibbs' theorem and partial molar volume
In the chemical engineering text of Smith, VanNess, and Abbott, there is a section on partial molar volume. It states that Gibbs theorem applies to any partial molar property with the exception of volume. Why is volume different? In other words, when evaluating the partial molar volume of a...- kayan
- Thread
- Replies: 4
- Forum: Materials and Chemical Engineering
-
P
Temperature and non-equilbrium thermodynamics
Suppose we have a body that isn't a black body, but has an arbitrary emission spectrum. In the general case, can the radiation from this body be characterized as having a temperature, and if so, how? If not, what conditions are needed to make the concept of temperature meaningful in this case?- pervect
- Thread
- Replies: 10
- Forum: Thermodynamics
-

Thermodynamics question about glass (quartz)
- Romantic_night
- Thread
-
- Tags
- Glass Quartz Thermodynamics
- Replies: 2
- Forum: Advanced Physics Homework Help
-
R
Question about the first law of thermodynamics
In thermodynamics, the internal energy (U) is the function of the volume (V) and temperature (T), U = U (T). Therefore, according to the mathematics, dU = π dV + Cv dT, where π is internal pressure and Cv is the constant volume heat capacity. Meanwhile, according to the 1st law, dU = δ w + δq...- RedCliff
- Thread
-
- Tags
- First law Law Thermodynamics
- Replies: 3
- Forum: Thermodynamics
-

Why is the change in entropy of a heat pump in one cycle equal to zero?
as it is not ideal, total change of entropy > 0 entropy loss of hot reservoir > entropy gain of cold reservoir why would the change in entropy of the heat pump in one cycle equals to zero? thank you- yecko
- Thread
-
- Tags
- 2nd law Law Thermodynamics
- Replies: 18
- Forum: Introductory Physics Homework Help
-

Temperature change brought about by radiation
I tried modeling the problem quite a few ways. one was to say that the difference between the heat emitted by the room and the heat emitted by the bulbs would equal the heat absorbed by the room, and that could allow us to calculate temperature. This did not work. I'd appreciate your help- Sai Maurice
- Thread
- Replies: 23
- Forum: Introductory Physics Homework Help