- #1
AngelLestat
- 25
- 5
- TL;DR Summary
- Radiant cooling loop between 15 c water lake and radiant floor installation.
Estimative calculation of the Copper or plastic heat exchange loop in length and flow speed.
This is a project that I wanted to do in the past 3 years, but I never was sure enough because I don't know how to calculate thermal transfer in a submerged coil to at least have an approximation of what to expect.
I want a cheap solution to pass the summer days without cook myself or wasting a lot of energy in the process.
I have the fortune to have a lake at the end of my yard, I measure the water temperature at its deepest point, which was 15 c at 10 meters deep, but a place of 8 meters deep is much closer and according to my measures is also between 15 c and 16 c (these measures were taken passing mid summer last year).
Is not a surprise, because cold temperatures of the winter reach the bottom of the lake, which by stratification stay there until the next winter because sunlight does not reach that depth.
I want to exploit those cold temperatures to cool my house in summer, my house already has a radiant floor in all the rooms which is used with a boiler to heat the house in winter.
I want to add the cool loop to the collector with an extra pump, I will hire a professional for the connection, but I want to make and install the Polypropylene pipe and a cooper loop myself (or PVC if there is no other choice).
Some diagrams:
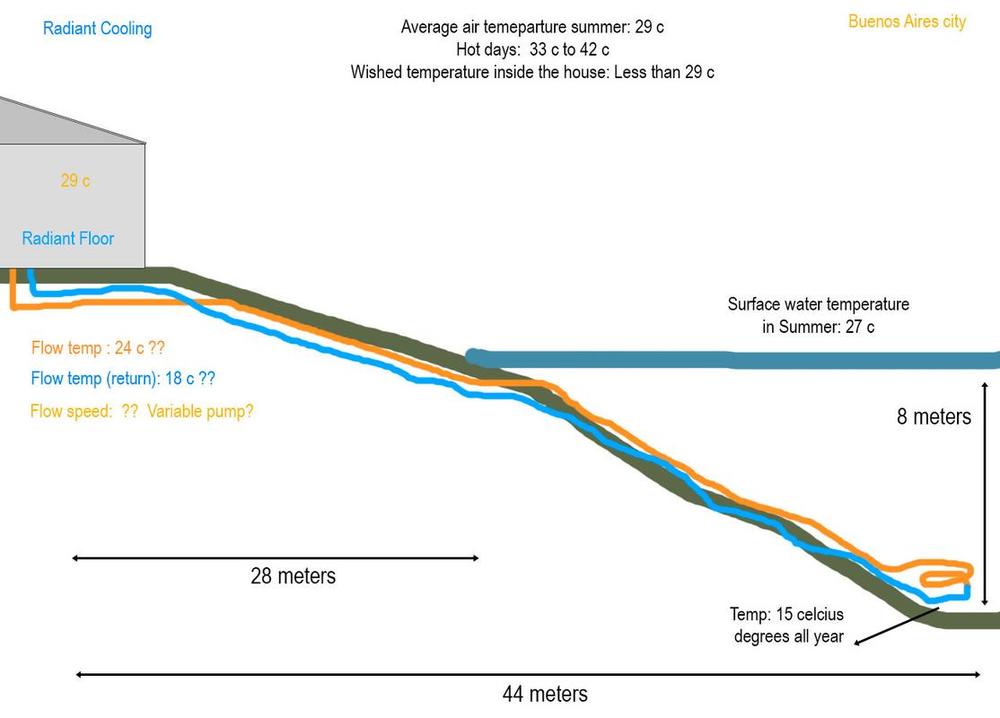
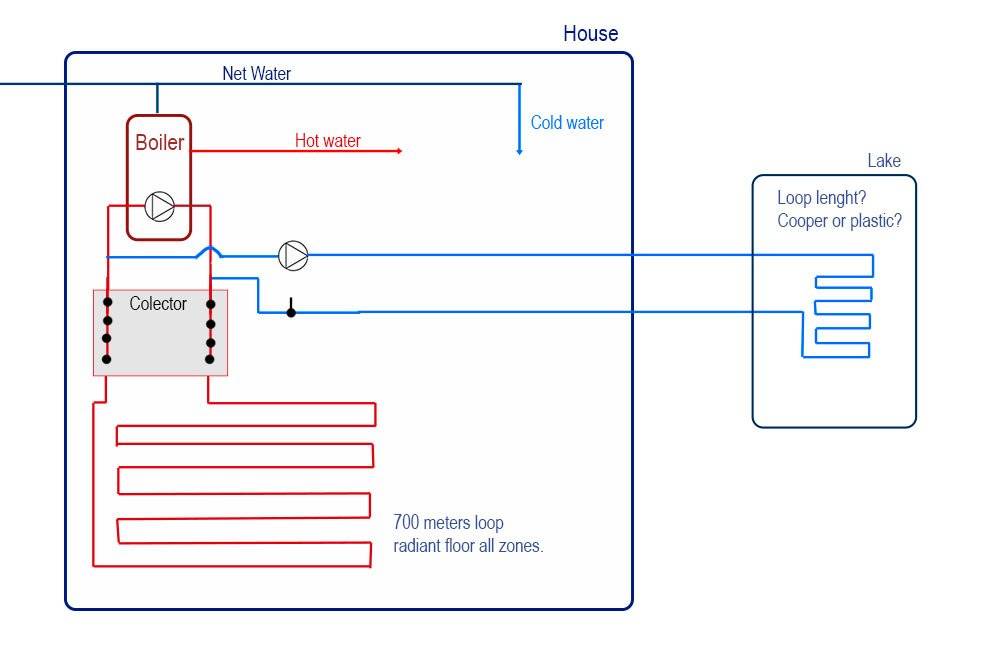
I want to use 1´ 1/4 of polypropylene black hose (the one that it is used for automated irrigation) to connect the collector to the loop in the lake (to avoid energy lost by friction), it would be thermal insulated with an extra layer and 20 cm buried until it reach the lake.
I have doubts about the material of choice for the loop, I know cooper is the best thermal conductor, but not sure about its corrosion, because water samples that I took from the lake bottom has a bit of rotten egg smell, which could mean hydrogen sulfide which in fact can corrode cooper (I guess). Another issue with cooper is that (if corrodes), high levels of copper sulfate could be bad for aquatic life, but some levels of copper sulfate are used for algae control, I made some math, and even if all my loop stays trap down there and dissolves (not my intention), it would be still 2 order of magnitude lower than the recommended level (I can add those calculations if you want).
The problem with polypropylene loop, is that it looks ugly due how massive it is, which can be seen in this picture https://plasticpipe.org/building-construction/12-geo_files/csss_images12/12geo.jpg
Then we have the fact that plastic is also polluting.
But those polypropylene loops are used to extract heat from ponds that are not deep with the help of a heat pump, so I am not sure how long they would need to be for my special case.
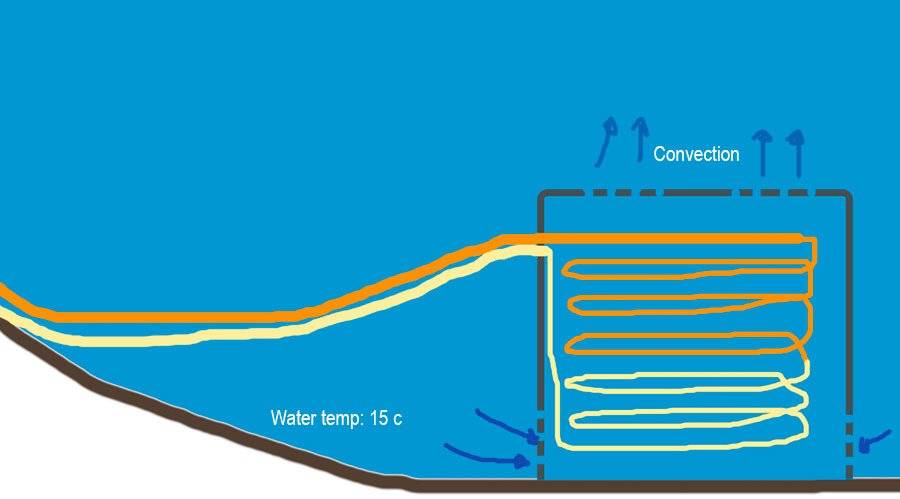
I can't do the math, but my common sense tells me that 2 copper loops in parallel of 3/4 by 10 meters each, could be enough for what I want, of course that is a total blind guest, because I am not sure how fast would be the cold water natural convection from the lake touching the copper pipes in order to cool them.
I will enclose the cooper loop in a cylinder tank with holes to avoid any fishing line stuck, but nobody fish there anyways.
So.. the question is.. Somebody knows how to approximate the length needed to accomplish at least 29 c inside the house on days of 40 c?
I was searching for some tables on heat transmission for submerged cooper or plastic loops, but I did not find nothing.
I understand that if I achieve 29 c inside my house, I will still have all the latent heat from moisture, and I also know that if the moisture is high the dew point could be reached which the floor would start to condense water.
But those problem seem easier to manage, because I can control the pump speed and I also have a medium AC unit for the living room of my house to help to reduce air moisture. I also have ceiling fans in many parts of my house which can help to vent hot air over the floor to maximize efficiency.
Something that I know for sure, that all that thermal mass at such cold temperatures just outside my house, looks like a waste opportunity when I am being cooked by floors, furnitures and walls at 40c, with no object to absorb that heat.
I want a cheap solution to pass the summer days without cook myself or wasting a lot of energy in the process.
I have the fortune to have a lake at the end of my yard, I measure the water temperature at its deepest point, which was 15 c at 10 meters deep, but a place of 8 meters deep is much closer and according to my measures is also between 15 c and 16 c (these measures were taken passing mid summer last year).
Is not a surprise, because cold temperatures of the winter reach the bottom of the lake, which by stratification stay there until the next winter because sunlight does not reach that depth.
I want to exploit those cold temperatures to cool my house in summer, my house already has a radiant floor in all the rooms which is used with a boiler to heat the house in winter.
I want to add the cool loop to the collector with an extra pump, I will hire a professional for the connection, but I want to make and install the Polypropylene pipe and a cooper loop myself (or PVC if there is no other choice).
Some diagrams:
I want to use 1´ 1/4 of polypropylene black hose (the one that it is used for automated irrigation) to connect the collector to the loop in the lake (to avoid energy lost by friction), it would be thermal insulated with an extra layer and 20 cm buried until it reach the lake.
I have doubts about the material of choice for the loop, I know cooper is the best thermal conductor, but not sure about its corrosion, because water samples that I took from the lake bottom has a bit of rotten egg smell, which could mean hydrogen sulfide which in fact can corrode cooper (I guess). Another issue with cooper is that (if corrodes), high levels of copper sulfate could be bad for aquatic life, but some levels of copper sulfate are used for algae control, I made some math, and even if all my loop stays trap down there and dissolves (not my intention), it would be still 2 order of magnitude lower than the recommended level (I can add those calculations if you want).
The problem with polypropylene loop, is that it looks ugly due how massive it is, which can be seen in this picture https://plasticpipe.org/building-construction/12-geo_files/csss_images12/12geo.jpg
Then we have the fact that plastic is also polluting.
But those polypropylene loops are used to extract heat from ponds that are not deep with the help of a heat pump, so I am not sure how long they would need to be for my special case.
I can't do the math, but my common sense tells me that 2 copper loops in parallel of 3/4 by 10 meters each, could be enough for what I want, of course that is a total blind guest, because I am not sure how fast would be the cold water natural convection from the lake touching the copper pipes in order to cool them.
I will enclose the cooper loop in a cylinder tank with holes to avoid any fishing line stuck, but nobody fish there anyways.
So.. the question is.. Somebody knows how to approximate the length needed to accomplish at least 29 c inside the house on days of 40 c?
I was searching for some tables on heat transmission for submerged cooper or plastic loops, but I did not find nothing.
I understand that if I achieve 29 c inside my house, I will still have all the latent heat from moisture, and I also know that if the moisture is high the dew point could be reached which the floor would start to condense water.
But those problem seem easier to manage, because I can control the pump speed and I also have a medium AC unit for the living room of my house to help to reduce air moisture. I also have ceiling fans in many parts of my house which can help to vent hot air over the floor to maximize efficiency.
Something that I know for sure, that all that thermal mass at such cold temperatures just outside my house, looks like a waste opportunity when I am being cooked by floors, furnitures and walls at 40c, with no object to absorb that heat.
Last edited: